Appendix 13 workshop 1
The Caspian sea, by virtue of the geographical position, isolation from the World ocean, specifics of water salt composition and salt content, other leading oceanological characteristics and composition of the biota represents a highly productive unique ecosystem. One of the major distinctive features of its biota is a small biodiversification, by the number of hydrobionts the Caspian ecosystem yields to all the seas of Russia (Zenkevitch, 1963; Eisarav, Yablonskaya, 1994). The high biological efficiency of the basin is formed due to the fact that high-energy ecological niches are occupied by 1-2 or several species rather close biologically. At the same time this factor creates an extraordinary vulnerability of the Caspian ecosystem, the successions (natural or anthropogenic) of which, affecting even one niche, have far-reaching consequences, defined by the length of the food chain and intensity of the attacking factor. For this reason the intrusion of new exotic species capable of preserving a high reproductive biological potential in the Caspian conditions, may cause a real eco-catastrophe.
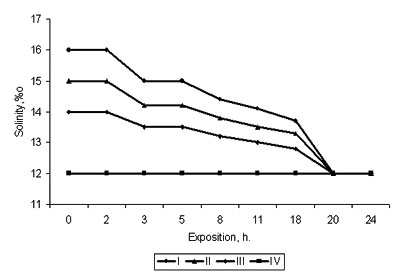
Fig. 1. Change of salinity at acclimation of Beroe ovata to the Caspian sea water
(I, II, III, IV - tests; initial salinity 18 ‰)
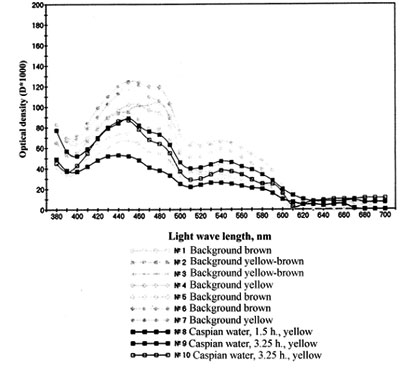
Fig. 2. Spectrum of the absorption by pigments of Beroe ovata in the Caspian sea water (N0 8, 9, 10)
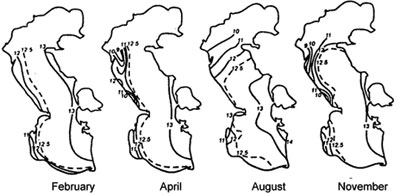
Fig. 3. Average salinity of upper level of the water column in the Caspian sea (from Zenkevitch, 1963)
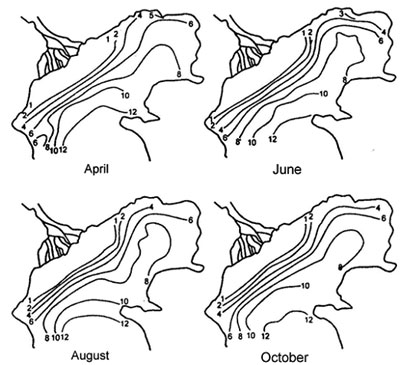
Fig. 4. Average salinity in the northern region of the Caspian sea (from Zenkevitch, 1963)
Unfortunately, such an event was marked with the intrusion of the ctenophore Mnemiopsis leidyi (A.Agassiz) in 1999 (NEP, 2001). Already in 2000 this intruder became mass organism and called negative and vast breakings in the pelagic zone of the basin. Under the data of KaspNIIRKH, by the end of 2000 the Caspian sprat had rather low (40-60 % from usual) rates of accretions and dimensionally - mass parameters by age-groups, and their total stock reduced more than twice. Already at the beginning of 2001 the mass destruction of sprat and seals was marked, and by results of the first half-year catches of sprat, herring, grey mullet, seals appeared in times, tens and hundreds of times less, than it was expected under the forecast, designed without taking into account the intrusion of Mnemiopsis and its affect on the biota (data of the Caspian scientific - fisheries council, Elista, August 23-24, 2001). The experience of examinations of biology and consequences of its intrusion in the Azov -Black sea basin (Ctenophore …, 2000) allows to predict:
1) the consequences of the intrusion of mnemiopsis in the Caspian sea as a whole in due course will be rougher, than it was marked in 2000-2001, since the existing food chain of the pelagic zone based on the zooplankton and sprat, as predators of the 1-st order, will be completely destroyed;
2) stocks of sprat, other fishes, which consume the zooplankton and sprat, will sharply (probably, by order and more) decrease, the behaviour of fishes in the traditional period of formation of accumulations for fishery will change;
3) catches of fishes will decrease sharply, both because of stock reduction, and change of conduct. Thus the efficiency of used implements of haul becomes low- they won’t be applicable in fishing.;
4) it is easy to imagine the scopes of economic losses and social problems of Caspian countries, which will arise at a practically complete degradation of the fisheries.
The similar scenario was already marked at intrusion of Mnemiopsis in the Azov sea, which one 1 year after its intrusion provoked the degradation of anchovy and tyulka sprat fisheries and of the bound with infrastructure, for the catches of these fishes made up 12-15 th t., i.e. decreased almost by 145 th t. as contrasted to the preceding 25-year period. 5 years after the intrusion of Mnemiopsis in the Black sea commercial catches losses of pelagic fishes exceeded 600 th. t/y., the damage of only catch losses were estimated in 240-340 million US dollars (FAO Fish. Rep, 1994), the Azov annual losses were estimated by a level of 40-45 million US dollars (Ctenophore …, 2000). Unfortunately, the Black sea countries selected the strategy “of doing nothing” concerning the ctenophore-intruder, except Russia, which State fishery committee by forces of AzNIIRKH organized regular and intensive monitoring of its population in the Azov-Black sea basin. Meanwhile, at the beginning of the 90-s substantial measures against the ctenophore-mnemiopsis and the damage, caused by it were offered. For this purpose it was recommended to utilize first of all another ctenophore - Beroe ovata, which accommodates itself to feeding only with other ctenophores (Mnemiopsis, Bolynopsis, Pleurobachia), and also some fishes, whose ration by 70-100 % consists of gelatinous organisms (Harbison, Volovik, 1993). These recommendations were supported by the UN experts (GESAMP, 1997).
In 1997 in the Black sea there was marked a new intruder-ctenophore Beroe ovata (Konsulov, Kamburska, 1998), and in 1999-2001 its explosive number in the north-eastern and other districts of the Black sea was registered. At once from the first year of Beroe development, the tendencies to enriching the state of the zooplankton and the complex of pelagic fishes of both Black and Azov seas (Ctenophore ..., 2000, Mirzoyan, Volovik, Martinyuk, 2001). The seminar organized within the framework of the Caspian ecological program (Baku, April, 2001) at which the leading scientists dealing with ctenophore studying in different seas of the World ocean, took part, recognized that in the existing circumstances Beroe ovata had the greatest preference at biocontrol (read - depressing) of development of the population of Mnemiopsis in the Caspian sea (CEP, 2001).
AzNIIRKH, resting on financial support of State fishery committee of Russian Federation, in 2000-2001 carried out a series of examinations of Beroe biology in the Azov and Black seas, assessment of its effect on the development here of the Mnemiopsis population, as well as executed a series of experimental works on studies of the tolerance of Beroe to environmental factors (salinity of water, temperature, nutrition, gas condition etc.), its acclimatization and bioproductional potential. The carried out works confirm, that Beroe ovata can successfully inhabit the area of the southern, central and the southern part of the northern districts of the Caspian sea, live and propagate here, consuming only ctenophore Mnemiopsis. Thus reproductive potential of Beroe is close to that of Mnemiopsis, and its tolerance to environmental factors – is similar to the corresponding parameters of Mnemiopsis, i.e. Beroe will dwell practically in the same places, as Mnemiopsis. In other words, this species may be a "magic wand", which largely (but not completely) will settle ecological, economic and social problems arisen with the intrusion of Mnemiopsis into the Caspian sea.
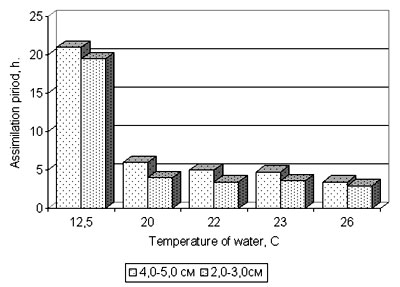
Fig. 5. Time of food assimilation by Beroe ovata specimens of different sizes in the Black sea water under temperature variations
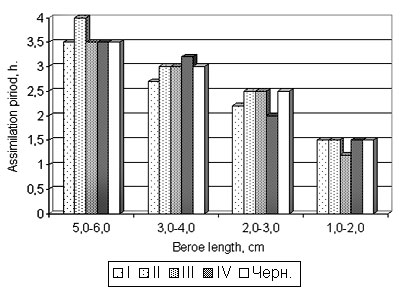
Fig. 6. Time of food assimilation by Beroe ovata during acclimation to the Caspian sea water
The note: the sizes of Mnemiopsis at feeding are commensurable to those of Beroe; temperature of water 26°N; I, II, III, IV – series of experiments

Fig. 7. Adaptation of Beroe ovata to lower temperature conditions with using of the Black sea water
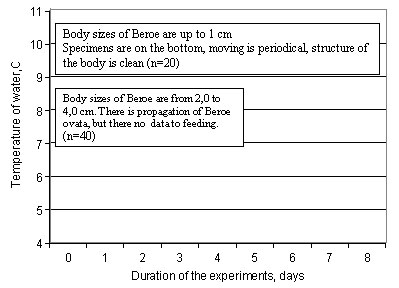
Fig. 8. Adaptation of Beroe ovata to lower temperature condition with using of the Caspian sea water
Let's consider the concrete factors and reasons for the benefit of Beroe ovata intrusion in the Caspian sea with the purposes of suppression the development of Mnemiopsis.
1. Reaction to salinity of water. In 1999-2001 there was a series of experiments on Boer’s reaction to water with salinity, equal to salt concentrations water in the Black, Azov and Caspian seas. Partially the outcomes of these works for 1999 are given in the publication of T. Shiganova with the co-authors (2000). The facts of Beroe habitation in surface layers of the water column, including the northwestern part of the Black sea, where the salinity compounded 10-14 ‰ (Saitzev, 1998), i.e. in a range from 18 ‰ near Turkey shores up to 10 ‰ near Odessa, confirm that this salt content with the composition which is practically identical by ratio components to waters of the World ocean (Zenkevitch, 1963), is favorable for this ctenophore. Both at sharp transmission of Beroe from the Black sea water with salinity of 18 ‰ into the water with salinity of 10,5 ‰, and at continuous decrease of salt content to the specified level, Beroe (small, medium and large specimens) behave uniformly, preserving typical motive and alimentary activity, make the process of reproduction. This fact is marked in the Azov sea, where Beroe propagated in Kuban districts (salinity 12-11 ‰). Therefore, the salinity up to 10,5 ‰ may be concerned as favorable for Beroe. At a further drop of salinity of water in aquariums up to 4,5 ‰ Beroe experiences signs of depression especially intensified at salinity less than 7,5 ‰. Salinity of 7,5 ‰ can be recognized by the lower level of saline tolerance of Beroe in water with salt composition, close to that of typically ocean waters.
The experiments carried out on keeping Beroe in water, by salt composition similar to the waters of the Caspian sea (artificially prepared on the basis of the Black sea water, and natural water from Central part of the Caspian sea), have shown, that Beroe in the Caspian water feels satisfactorily, preserving a typical motive activity, feeding behavior and propagating, and the produced eggs passed embryogenesis, the conforming stages of ontogenesis (tab. 1, fig.1). At sharp transmission of Beroe from the Black sea water (18 ‰) in the Caspian water (12 ‰) at first the animals felt depressed (about 3 hours) (lied on the bottom of aquarium, changed coloring, with their cilia actively moving), then they recovered and did not differ from the specimens being as a control samples (tab. 1). The fact that all functions of Beroe vital activity in the Caspian water were restored within 3-3,5 hours is confirmed by regeneration of light absorption spectrums by pigmentary spots up to a level of background parameters (fig. 2). Minimal salinity of the Caspian water in experiences at various temperature, at which the animal in experience felt satisfactorily, was 6,5-7,5 %i Therefore, by saline conditions Beroe can inhabit the southern, central and southern part of the northern district of the Caspian sea (fig. 3,4).
2. Feeding behavior, duration of nutrition assimilation depended on Beroe sizes, but did not depend on that fact, whether these phenomena occurred in the Black sea or Caspian water (tab. 1, fig. 5,6).
3. Beroe reacted similarly to the ambient temperature both in Black sea, and in the Caspian water. With temperature rise activity and vital functions increase (fig. 5,6). The highest parameters are marked at water temperature of 20° C and more, and the processes are approximately identical. However lower temperature of water Beroe can both feed, and propagate in the conditions of the Black and Caspian sea (see fig. 7,8). Thus, Beroe can dwell in the surface layer of the water column of all the Caspian sea, except its extreme northern and north-eastern districts (fig. 9, 10).
4. Beroe preserved similar intensity of oxygen consumption at vital activity (fig. 11,12) both in the Black sea, and the Caspian sea waters at different temperature. Under the contents of oxygen the upper strata of the Caspian waters (fig. 13) will be accessible for Beroe.
5. It was not possible for us to estimate the fecundity of Beroe in the Caspian waters. But in the Black sea water fecundity of Beroe of different sizes varied from 0,7 to 10,7 thousand eggs, within 2-3 days the specimens produce 3,7-18,3 thousand eggs at sufficient nutrition (tab. 2,3). Beroe is likely to preserve such fecundity level in the Caspian waters. It is necessary to stress, that these parameters are close to evaluations of fecundity of Mnemiopsis (Pianka, 1974, Shiganova, 2000), and acknowledges the thesis about similar and high bioproductional potential of both considered species.
Table 2. Fecundity of Beroe ovata in the different experimental conditions (t=230C)
|
Conditions of experiments |
Body sizes of Beroe, cm |
Total number of eggs per one specimen |
|
the darkness + the hunger |
7,6x4,6 |
10680 |
| darkness + feed (1 M.l* 3o4 cm) |
7,6x4,4 |
10773 |
| 6 h. by the darkness + the feeding (larva of M.l.) |
6,0x5,5 |
1444 |
| 12 h. by the darkness + the feeding (larva of M.l.) |
6,0x5,5 |
722 |
| 24 h. by the darkness + the feeding (larva of M.l.) |
6,5x5,5 |
2527 |
| 24 h. by the light + the feeding (larva of M.l.) |
6,1x5,3 |
1800 |
| 24 h. by the light + the feeding (M.l. 4o3,3 cm) |
5,8x4,8 |
1560 |
| The feed – freezing M.l. |
3,4x2,9 |
7025 |
| T=90C |
6,7x4,8 |
48 |
*Mnemiopsis leidyi
Table 3. Fecundity of one specimen of Beroe ovata (total number of eggs per 1 specimen per day) (t=230C)
|
NN |
1 day |
2 day |
3 day |
Conditions of experiments |
|
1 |
7680 |
10777 |
- |
darkness |
|
2 |
2520 |
10650 |
4350 |
|
|
3 |
9120 |
1800 |
the light + the feeding (M.l. 4o3,3 cm) |
|
|
4 |
2040 |
1680 |
the light + the feeding (larva M.l.) |
6. Both known literary data (Kamshilov, 1961; Swanberg, 1974) and our publications (Ctenophore…, 2000) as well as newly-obtained data confirm, that Beroe is the obligate consumer only of other ctenophores. That means, that no damage will be caused to other representatives of the Caspian plankton, including eggs and larvae of fishes, excluding Mnemiopsis, which will be intensively eaten away. It is necessary to mention that Beroe can mechanically swallow other organisms, but this phenomenon, in our observations, happens very seldom, in single cases and incidentally swallowed organisms are vomited fast and without damages.
Making a comparative evaluation of distribution and biology of Beroe, its affect on Mnemiopsis in the Black sea with the data on these aspects in the Caspian and its possible habitat in the Caspian sea one may come up to the conclusion sea specified possible (probable) geographic ranges it ctenophore in Caspian sea, that they do not have essential differences. Beroe can successfully inhabit, like Mnemiopsis, the upper strata of the water column almost of all the area of the Caspian sea. Without concerning co- habitation of Mnemiopsis and Beroe in the Azov-Black sea basin, we consider it important, that the latter reduced the number of Mnemiopsis in rather short time after the intrusion and improved (but not restored completely) the course of biological processes in the pelagic zone of the basin. Similar situation may be expected and in the Caspian sea, under the condition of the intrusion there of Beroe ovata.
The works carried out in the Azov-Black sea basin allowed to formulate the idea and contents of measures, the realization of which will allow to practically completely neutralize the effect of Mnemiopsis on the ecosystem of the seas, but these problems demand some more further scientific elaboration.
Thus, the modern knowledge about ctenophores Mnemiopsis and Beroe in their new geographic range allows to make an only conclusion, that the intrusion of Beroe ovata in the Caspian sea will mitigate the problems created by Mnemiopsis and will not cause any damage and aggravation of the ecosystem of the Caspian sea.
References
Ctenophore Mnemiopsis leidyi (A.Agassiz) in the Azov and Black seas: biology and consequences of the intrusion // Under scientific edition by prof. S.P. Volovik. – AzNIIRKH, Rostov-on- the Don, 2000. - 500 p.
Kamshilov N.M. A biology ctenophore of the Murman littoral // Publications of the Murmansk marine biological institute.- 1961.- Edition. 3 (7).
Mirzoyan I.A, Volovik S.P., Martinyuk M. Features of development of the population of Beroe ovata in the Azov-Black Sea basin and consequences of its intrusion // Publications. The Caspian buoyant university, 2001, in printing.
Shiganova T.A. Some conclusions on studies of intruder Mnemiopsis leidyi (A.Agassiz) in the Black sea. In the book "Ctenophore Mnemiopsis leidyi (A.Agassiz) in the Azov and Black seas: biology and consequences of the intrusion // Edit. Prof. S.P. Volovik. – AzNIIRKH, Rostov-on- the Don, 2000. - P. 33-75.
Shiganova T.A., Bulgakova Y.V., Volovik S.P., Mirzoyan Z.A., Dudkin S.I. New intruder Beroe ovata and its affect on the ecosystem of the Azov-Black Sea basin in August, - September 1999, p. 432-449.
Zenkevitch L.A. Biology of the seas of the USSR. // M.: From - in SA USSR, 1963.- 739 p.
Harbison G.R. and Volovik S. P. Methods for the Control of the Ctenophore Mnemiopsis leidyi in the Black and Azov Seas. FAO Fisheries Report No 495. Rome, 1994, p. 32-44.
Harbison G.R. and Volovik S.P. The ctenophore Mnemiopsis leidyi in the Black sea. In: Nonindigenous Estuarine and Marine Organisms (MRMO). 1994, p. 25-26.
CEP 2001 - Caspian Environmental Program, Mnemiopsis workshop (Baku, 24-26 April 2001) final report; CEP Wb site: http://www.http://www Caspian environment. org/biodiversity/meeting. htm
GESAMP. Report for the Meeting UNEP/IMO/FAO/UNESCO. Second Meeting of the GESAMP Working Group on Opportunistic Settlers and the Problem of the Ctenophore Mnemiopsis leidyi in the Black Sea. Draft 2. Geneva. 20-24.03.95, 1995.- Published 1997. - 65 p.
Konsulov A., Kamburska L. Ecological determination of the new Ctenophore - Beroe ovata invasion in the Black Sea // Tr. Ins. Oceanology BAN. - Varna, p. 195-197.
Kosarev A.N., Yablonskaya E.A. The Caspian Sea. SPB. The Haque. 259 p
Pianka, H.D. Ctenophora. In: Reproduction of Marine Invertebrates, (A.C. Giese and J.S. Pearse, eds.), Academic Press, N.Y. Vol. I, 1974, p. 201-265.
Swanberg N. The feeding behavior of Beroe ovata. Mar. Biol. 24. 1974, p. 69-76.
Table 1. Adaptation of Beroe ovata to the different salinity*
| Terms of observation, hours |
Tests |
||||
|
I |
II |
III |
IV |
||
|
Beroe size groups (cm) |
|||||
|
4. 1,0-1,5 |
4. 1,0-1,5 |
4. 1,0-1,5 |
||
| 0** | Beroe are placed in the aquariums with corresponding salt concentrations | ||||
| 0,5 |
Specimens changed coloring, descended to the bottom of the pot, locomotion of lobe plates is active |
Specimens descended to the bottom at once, changed coloring, all are alive, locomotion of lobe plates is active |
|||
| 2 |
Move in the water, locomotion of paddle plates is active |
Start to move, to lift in the water |
|||
| 3 |
The behavior did not change. Adapted Mnemiopsis specimens of different sizes are placed in the pot, Beroe demonstrate a typical feeding behavior |
All are alive, lift in water strata, the restore the initial color | |||
| 5 | The feeding behavior has not change: at contact the immediate seizure of a victim is marked |
do not feed, but lift up actively enough: descend to the bottom and ascend to the upper layers |
Specimens of the 1 group capture Mnemiopsis |
||
|
Specimens of groups 2 and 3 are more active |
Specimens of group 4 are more active |
Specimens of groups 2 and 3 began to feed |
|||
| 8 |
The food is digested specimens of groups 2 and 3 swallow a new Mnemiopsis |
Mnemiopsis is observed in the gastrovascular cavity of the majority of specimens |
|||
| 11 |
Specimens of groups 3 and 2 are more active |
All are active. Mnemiopsis is observed in the gastrovascular cavity of the majority of specimens |
Mnemiopsis is observed in the gastrovascular cavity of the majority of specimens |
Large specimens from the group digest food, small specimens from the 4th group –"attack" the large Mnemiopsis |
|
| 18 | Locomotion and alimentary behavior without changes |
Feed actively, all the Mnemiopsis specimens are eaten |
|||
| 20 |
All the animals are placed in the Caspian sea water |
||||
| 24 |
All alive. Locomotion and alimentary behavior without changes. |
||||
* changes of salinity are given in fig. 1
** Terms of dilution of the Black sea water with Caspian water
Fig. 9. Average temperature (0C) of the upper level of the water column in the Caspian sea (from Zenkevitch, 1963)
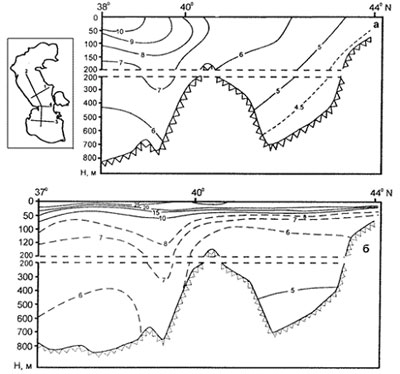
Fig. 10. Temperature of water column (transection by meridional direction) in February (a) and August (b) (from Kosarev, Yablonskaya, 1994)
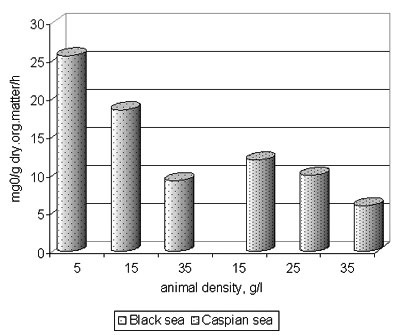
Fig. 11. Velocity of oxygen consumption by Beroe ovata in the Black sea and Caspian sea waters in dependence on gravity animal in a unit water volume (temperature 18°N)
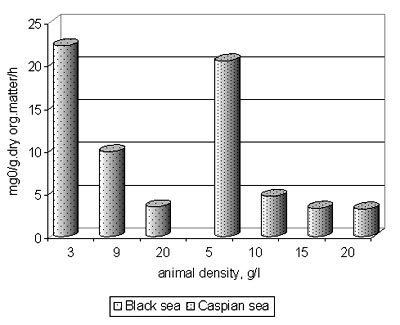
Fig. 12. Velocity of oxygen consumption by Beroe ovata in the Black sea and Caspian sea water in dependence on animal density in a unit of water volume (temperature28°N) (n=6)
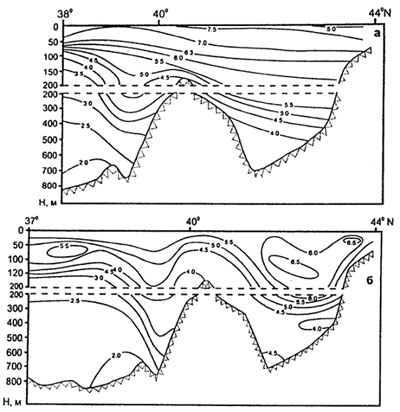
Fig. 13. Dissolved oxygen contains (mg/l) (transection by meridional direction) in February (a) and August (b) (from Kosarev, Yablonskaya, 1994)
Document Actions
