A Report
Prepared for the Caspian Environment Program, Baku, Azebaijan
in December-2001
Ahmet E. Kideys & Tamara
Shiganova*
Institute of Marine Sciences, Erdemli, Turkey
*Shirshov Institute of Oceanology, Moscow, Russia
1. ABSTRACT
During the conference devoted to Mnemiopsis, invader of Caspian Sea, held in Baku,
Azerbaijan, 24 – 26 April 2000, the working group has suggested producing a document
describing methods for monitoring Mnemiopsis and other gelatinous macroplankton in the
Caspian Sea in order to evaluate their impact on the ecosystem. In this document three
types of data collection were suggested to be deployed; these being (1) the basin-wide
sampling, (2) coastal monitoring and (3) sightings by volunteers. The document includes
detailed sampling and processing methods, too.
2. INTRODUCTION
Mnemiopsis which was brought in 1980s from coastal waters of the northern America
to the Black Sea caused tremendous damage in this new ecosystem (Vinogradov et al, 1989,
1992, Shiganova,1997, 1998; Kideys 1994; Kideys et al. 2000. This ctenophore have now
appeared in the Caspian Sea since 1997-1998. At the time of Mnemiopsis invasion,
there was also found jellyfish Aurelia aurita in the Caspian Sea, permanent
resident of the Black Sea (Ivanov et al, 2000, Shiganova et al, 2001).
By the year 2000 a substantial increase in Mnemiopsis
biomass, particularly for the Southern and Mid Caspian was noted with a decline in the
biomass of mesozooplankton which adversely changed the nourishment of sprats (Shiganova et
al. 2001). Similar results were obtained from the monitoring programs set up in two
different coastal regions in the Iran and Azerbaijan coasts of Caspian (Kideys et al.
2001a,b).
During the conference devoted to Mnemiopsis,
invader of Caspian Sea, held in Baku, Azerbaijan, 24 – 26 April 2000, the working
group has decided to produce a document for “Methods for monitoring Mnemiopsis
and other gelatinous macroplankton in the Caspian Sea” for the assessment of their
impact on the Caspian ecosystem. Therefore this document has been prepared to guide
studies relating gelatinous organisms in the Caspian Sea using a similar methodology.
Should you have any questions regarding
this document please do not hesitate to contact Tamara Shiganova (shiganov@chip.sio.rssi.ru) and Ahmet
Kideys (kideys@ims.metu.edu.tr).
3. IDENTIFICATION OF GELATINOUS
ORGANISMS
3.1. Mnemiopsis
Mnemiopsis (Fig.1) is a lobate ctenophore with two large oral lobes,
transparent and a bit whitish. The animal moves by means of cilia on rowing membranes. The
cilia are directed towards aboral side, so that ctenophore swims with mouth ahead.
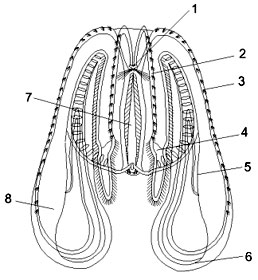
Fig. 1. Mnemiopsis leidyi. (1) aboral organ, (2) subtentacular row
of comb flappers, (3) subsagital row of comb flappers, (4) auriculus, (5) subsagital tube,
(6) translobal tube, (7) tentacular tube, (8) lobe (from Shiganova 2000).
The sizes of Mnemiopsis found in the
Caspian Sea are much smaller than those found in the Black Sea. For example during the
August 2001 cruise in the mid and southern Caspian, The maximum size of the ctenophore was
only 41-45 mm and the bulk of individuals (85.5%) were less than 10 mm in length (A.
Kideys & M. Moghim, unpublished data). Whereas the individuals found in the Black Sea
reach the size of 14-18 cm.
The eggs of Mnemiopsis are round,
0.15-0.3 mm in diameter. The larva has spherical form without lobes, with long catching
tentacules and has the size of about 0.15-0.20 mm (Fig 3 A, B); lobes began to evolve when
the individual exceeds 10 mm in length. (Fig 2).
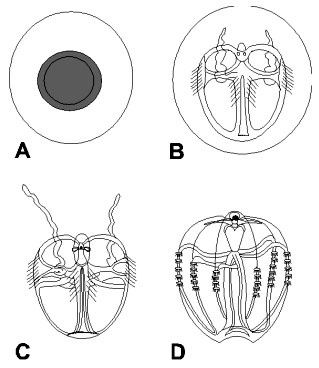
Fig. 2. Early life stages of Mnemiopsis. (A) newly laid egg, (B)
embryo with tentacles, about 30 hours old, still within the egg envelope, (C) newly
hatched embryo in cydippid stage, (D) embryo about 32 hours old showing muscular strands
(from Shiganova 2000).
3.2. Aurelia aurita
The jellyfish A. aurita has a complicated life cycle with several sessile and
pelagic stages - planula, scyphistoma, ephyra and finally medusa. The most remarkable and
recognizable stage is medusa (Fig 3).
 
Fig 3. Aurelia aurita.
Planula - larva stage stays in plankton for
about a week and then sink on the bottom. Scyphistoma develops from the sunken planula,
which has an appearance similar to polyp with 16-32 feelers. They usually occur in shallow
areas, in sublitoral, usually attached to oysters or mussels. Scyphistoma can germinate
another larval stage called ephyra which grows into medusa. A. aurita ephyrae has
the size of 3-6 mm and may appear in different times in different regions depending on the
temperature. Ephyrae appear between December and April in the Black Sea.
3.3. Other gelatinous macroplankton of
the Caspian Sea
Ctenophores Pleurobrachia pileus (Fig. 4) and Beroe ovata (Fig. 5) which
occur in the Black Sea may appear anytime in the Caspian. Other gelatinous of
macroplankton species of the Caspian Sea are hydrozoans Blackfordia virginica, B.
pallasi, Maeotica sp, Moerisia pallasi (Fig. 6), Bougainvillia megas, Perigonimus
megas.
Fig. 4. Pleurobrachia pileus
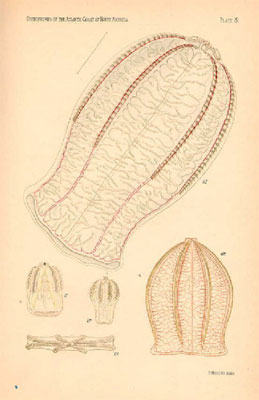
Fig. 5. Beroe cucumis sensu
Mayer, 1912 (# 67) and Beroe ovata sensu Mayer, 1912 (# 68 and 69). Note the
extreme flattening of Beroe ovata sensu Mayer, 1912 (# 69) (from R.
Harbison).
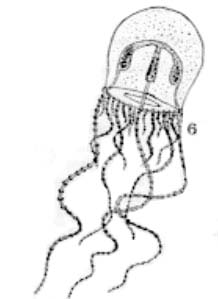
Fig. 6. Moerisia pallasi
4. TYPES OF MONITORING FOR MONITORING OF
MNEMIOPSIS AND OTHER GELATINOUS ANIMALS
Three types of monitoring are proposed:
- a) From basin-wide cruises
- b) From regular coastal monitoring at selected stations by
institutes or scientific establishments of riparian countries
- c) From observations by volunteers
a) Basin-wide cruises
Cruise Timing
It is reasonable to conduct cruises at least once in a season: in spring (March-April); in
summer (August - September); in fall (October-November); and in winter (December –
January). The most important sampling materials are obtained in August-September, when,
based on Black Sea experience, one can anticipate an intensive reproduction, and
consequently maximum abundance and biomass of Mnemiopsis population.
However, since seasonal dynamics of Mnemiopsis
in the Caspian Sea is not known yet, it is even more desirable to conduct studies on a
monthly basis.
Investigation of Spatial distribution
It is important to conduct studies in all parts of the Caspian due to differences in
temperature and salinity regimes as well as hydrochemical characteristics of the Northern,
Middle and Southern Caspian.
Vertical Distribution
The investigation of vertical distribution of Mnemiopsis should be implemented at
least at two stations (one shallower and one deeper) in each region of the Caspian making
use of a closing net. The important parameters to take into account should be the presence
of the thermocline and pycnocline; the layer of sampling could be adjusted depending on
the position of these layers.
At the first stage it is suggested
to sample the following vertical layers: 500-250m, 250-100m, 100-50m, 50-25m and 25-0m.
Occurrence of diel vertical migrations are determined by analyzing samples taken at day
and night time.
Additional sampling could be performed from
the thermocline to the surface, from pycnocline to thermocline. For sound results, each
layer should be sampled at least three times. A minimum of two stations (where Mnemiopsis
abundances are seemingly high) should be studied for this aim.
b) From regular coastal monitoring at
selected stations by institutes or scientific establishments of riparian countries
For monitoring it is desirable to have at
least 2-3 stations (optimally 10 stations). The distance between stations should be kept
as far as possible. If the institute or other research institution has long term stations
already established, they should be used for Mnemiopsis study.
Sampling Interval
It is recommended to take samples biweekly from early March to late November and monthly
for the rest of the year.
c) Observation by volunteers
The volunteers can be any inhabitants of coastal towns of riparian states. The best
candidates are University or Institutes students, fishermen and oil workers. Due to the
presence of only one ctenophore species in the Caspian Sea at present, volunteers could
easily categorize gelatinous organisms in two groups: 1) Mnemiopsis and 2) Other
gelatinous organisms. If a volunteer wants to be sure if it is a Mnemiopsis or
other gelatinous organisms, a scoop could be deployed. An example of “reporting
form” is given in Appendix 1.
5. SAMPLING AND SAMPLE PROCESSING
(To be used during monitoring a and b given above)
For the sampling of Mnemiopsis and
other gelatinous organisms it is recommended to deploy a net with wide diameter (=or
>50 cm) equipped with net material of 500 micron of mesh size. Information on some nets
to be used in monitoring studies are given in Appendix 2.
Sampling is performed by vertical hauls of
certain water column from the board of a vessel. The net is lowered by winch equipped with
length meter to measure the length of released rope (ropes diameter 4-6mm). The velocity
of lifting should be 1 meter per second. Depth of sampling should be corrected with taking
into account angle of rope in the process of hauling. In the case of small ship and small
depths, if the required equipment (winch, arrows) is absent, it is possible to perform
vertical hauls from the board by hands using a suitable net.
5.1. Measurement of Mnemiopsis
and other gelatinous organisms
Since the fixation of gelatinous macroplankton is very problematic, various measurements
of these organisms should be taken immediately.
Mnemiopsis
In order to measure the total (with lobes) and/or oral-aboral (without lobes) length,
ctenophores should be placed individually in a Petri dish with very small volume of water.
If there are <50-100 individuals in the sample; all Mnemiopsis will be sorted
out to size groups on Petri dish using a ruler and numbers will be counted as follows:
| Length Group (mm) |
Mnemiopsis number |
| 0-5 |
…. |
| 6-10 |
…. |
| 11-15 |
…. |
| 16-20 |
…. |
| 21-25 |
…. |
| 26-30 |
…. |
and so on..
Ctenophore’s weight could be
calculated by using length-weight equation obtained for Caspian Mnemiopsis as given
following (Kideys et al 2001b):
Weight (gr) = 0.0011 Length (mm)2.34,
R2=0.65, n=98. The total biomass is assessed as the sum of products of average
animal weights in the group and total number of individuals in the group.
If the sample size is not higher than 100
individuals, all animals should be measured individually, otherwise a subsampling could be
performed (1/2, 1/3, 1/4, etc. of total), Then a recalculation for the entire sample
should be executed.
The Mnemiopsis eggs and larvae
should be fixed in 2% buffered formaldehyde along with other mesozooplankton
Jellyfish Aurelia aurita
The jellyfishes can be grouped with 1 cm intervals and then the number of animals for each
size group is recorded. The jellyfish A. aurita can be fixed in 2% buffered
formaldehyde. We are proposing the following equation for calculation of biomass of A.
aurita, which was developed for A. aurita inhabiting in the Black Sea:
W= 0.12 L 2.58 where W- wet weight in grams, L- length in mm;
(measurements range from 6.1 to 151mm)(Mutlu, 2001)
These equations should also be developed
for the Caspian population of A. aurita.
5.2. Calculation of abundance and
biomass of gelatinous organisms
To assess the abundance and biomass it is
necessary to count total number of ctenophores or/and jellyfish in the sample, and then
assess total abundance and biomass for the area (i.e. per m2) and/or water
volume (per m3 by taking sampling depth into account).
Some investigators use coefficient for the
filtering efficiency of the net, whilst some others not. The Institute of Oceanology
Russian Academy of Sciences has adopted factor for the non-caught gelatinous organisms for
the sampling with BR net in relation to underwater observations (Vinogradov et al, 1989).
For Mnemiopsis
- < 10 mm, coefficient k - 2;
- 10-45 mm, coefficient, k -2 ;
- >45 mm, coefficient –2.3
For A. aurita:
- <50 mm, coefficient, k-2 ;
- 50-100 mm, coefficient, k-2.3;
- >100 mm, coefficient k-3.
If a correction factor is applied, this
should be clearly stated in the methods.
Other gelatinous organisms
In case of discovery of any unknown
gelatinous organisms, if possible one should take their photograph, measure, describe them
and try to preserve them in 2% buffered formaldehyde or glutaraldehyde.
5.3. Basic parameters to be measured
in course of monitoring
Hydrophysical parameters:
- Temperature (preferably at all depths or at least in the
surface waters).
- Salinity (preferably at all depths or at least in the
surface waters).
- Turbidity (Secchi depth).
Hydrochemical parameters:
- Nutrients and oxygen samples from selected depths down to
the sea bottom.
Biological parameters:
- Phytoplankton samples from standard depths (2 liter of
seawater from depths of 0, 10, 25, 50 m: for methodology see Kasimov 2000).
- Mesozooplankton (including ichthyoplankton) samples (using a
vertical net tow from 100 m to the surface for deep stations and from the bottom to the
surface for shallow stations). It will be appropriate to use a mesh size of 100 to 200 um
(see Kasimov 2000 for methodology) for zooplankton and 500 um for ichthyoplankton . The
most optimal net for zooplanlankton sampling is a Juday net and an ichthyoplankton net
which are available in the most Institutes of Caspian countries with mesh size equal to
125, 180 or 200 micron for a Juday net. Alternatively a METU Net (designed by Dr Ahmet
Kideys) with different mesh sizes of 112-200 micron could also be used. Again some
investigators may use coefficients for the filtering afficiency of mesozooplankton
(Shirshov Institutse’s coefficient is 2 for mesozooplankton). If any coefficient is
used, again, this should be clearly stated.
- In addition to phytoplankton samples, sampling for chl-a
from phytoplankton sampling depths.
The cruise data recording sheet prepared
for the Caspian coasts of Iran could be modified according to aim (see Appendix 3).
REFERENCES
Ivanov PI, AM Kamakim, VB Ushivtzev, T.
Shiganova, O. Zhukova, N. Aladin, SI Wilson, GR Harbison, HJ Dumont 2000. Invasion of
Caspian Sea by the comb jellyfish Mnemiopsis leidyi (Ctenophora). Biological
Invasions 2: 255-258.
Kasymov A 2000. Methods of monitoring in
Caspian Sea. Gapp-Poligraf Publ., Baku, pp. 26-32.
Kideys AE 1994. Recent dramatic changes
in the Black Sea ecosystem: The reason for the sharp decrease in Turkish anchovy
fisheries. Journal of Marine Systems 5: 171-181.
Kideys A. E., A. V. Kovalev, G. Shulman,
A. Gordina & F. Bingel 2000. A review of zooplankton investigations of the Black Sea
over the last decade. J. of Marine Systems, 24:355-371.
Kideys A.E. & Z. Romanova (2001).
Distribution of gelatinous macrozooplankton in the southern Black Sea during 1996-1999.
Marine Biology 139: 535-547.
Kideys A. E., Shahram Ghasemi, Davood
Ghninejad, Abulgaseem Roohi & Siamak Bagheri 2001a. Strategy for combatting
Mnemiopsis in the Caspian waters of Iran. A report prepared for the Caspian
Environment Programme, Baku, Azerbaijan, Final Report, July 2001.
Kideys A. E., F. M. Jafarov, Z. Kuliyev
& T. Zarbalieva 2001b. Monitoring Mnemiopsis in the Caspian waters of
Azerbaijan. A report prepared for the Caspian Environment Programme, Baku, Azerbaijan,
Final Report, August 2001.
Mutlu E. 2001. Distribution and abundance
of moon jellyfish (Aurelia aurita) and its zooplankton food in the Black Sea. Marine
Biology 138: 329-339.
Shiganova T.A. 1997. Mnemiopsis leidyi
abundance in the Black Sea and its impact on the pelagic community. In "Sensivity
of the North, Baltic Sea and Black Sea to antropogenic and climatic changes"
Sp.Volume. Ed. E. Ozsoy and A.Mikaelyan. Kluwer Acad. Pub.p.117-130.
Shiganova T.A. 1998. Invasion of the
Black Sea by the ctenophore Mnemiopsis leidyi and recent chanches in pelagic community
structure. Fisheries Oceanography –Globec Special Issue Ed.Steeve Coombs.p.305-310
Shiganova T.A.Kamakin A.M., Zhukova O.P.,
Ushivtzev V.B., Dulimov A.B.Musaeva E.I. Invader in the Caspian Sea ctenophore Mnemiopsis
and initial results of its impact on the pelagic ecosystem. 2001, Oceanology V.41, N 4.
Pages 542-549.
Shiganova T.A. Some results of
study of Mnemiopsis leidyi biology in the Black Sea // “Ctenophore Mnemiopsis
leidyi (A.Agassiz) in the Azov and Black Sea and consequences of its intrusion”.
Ed.S.P. Volovik. Rostov –on-Don. 2000 b:.pages 33-75.
Vinogradov M.E., E.A. Shushkina, E.I.
Musayeva, P.U.Sirokin. New invader in the Black Sea – ctenophore Mnemiopsis leidyi
(Agassiz) // Ocean Science 1989, V. 29, issue 2, pages 293-299.
Vinogradov M.E., V.V. Sapoznikov, E.A.
Shushkina. The Ecosystem of the Black Sea, M.Nauka, 1992, page 112.
Appendix 1
‘Mnemiopsis Reporting Form for Volunteers to obtain data on Mnemiopsis and
other gelatinous organisms’
Date:
Hour:
Area:
Volunteer’s name:
Volunteer’s occupation (please tick appropriate box):
- Student
- Fisherman
- Seaman
- Oil Rig Worker
- Other
Mnemiopsis:
- Less than 1-2 individuals in m3
- Between 3-10 individuals in m3
- More than 10 individuals in m3
- If possible then please indicate size composition and
peculiarities of spatial distribution
Other gelatinous organisms:
Short description:
Body shape:
Sizes:
Color:
Average quantity in m3
Comments:
Please forward this information to
...
Appendix 2
Some plankton nets recommended for big
boats (BB) and small boats (SB)
Type of net |
Diameter
(cm) |
Mesh Size
(um) |
Plankton
Group |
METU Net |
50 |
500 |
Macro (SB) |
Hensen Net |
70 |
300 |
Macro (BB) |
Bogorov-Rass
net (BR) -13/140 |
113 |
530 |
Macro (BB) |
Big cone Net |
80 |
530 |
Macro (BB, SB) |
Small cone Net |
50 |
~ 900 |
Macro (SB) |
Juday
net-80/113 |
80 |
180 |
Meso (BB) |
Juday net-37/50
|
37 |
180 |
Meso (BB, SB) |
Small Cone Net |
50 |
180 |
Meso (SB) |
METU Net |
50 |
100-200 |
Meso (SB) |
|
|
|
